
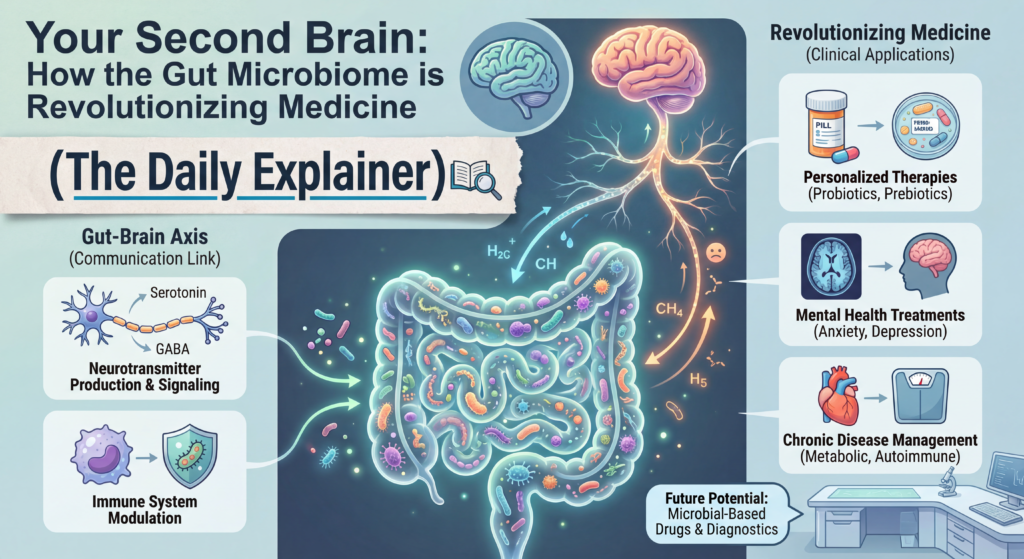
Visualizing the major communication highways linking your gut microbiome, brain, and immune system.
Introduction – Why This Matters
For centuries, medicine viewed the gut as a relatively simple tube: food goes in, waste comes out. Any complaints were localized—indigestion, ulcers, irritable bowel. Today, a seismic shift is underway. Cutting-edge research reveals your gut is not a passive pipe but a vibrant, living command center, communicating directly with your brain, tuning your immune system, and influencing everything from your mood to your risk for chronic disease. This is the Gut-Brain-Immune Axis, and understanding it is revolutionizing how we think about health.
The trillions of bacteria, viruses, and fungi that reside in your intestines—collectively called the gut microbiome—act as a vital organ. They produce essential vitamins, metabolize drugs, and, crucially, manufacture thousands of signaling molecules that travel through your bloodstream and nerve pathways. When this microbial community is diverse and balanced (a state called eubiosis), it promotes health. When it’s disrupted ( dysbiosis), it can contribute to a staggering array of conditions far beyond the gut: depression, anxiety, autoimmune diseases like rheumatoid arthritis and multiple sclerosis, obesity, type 2 diabetes, Alzheimer’s disease, and even how you respond to cancer immunotherapy.
In my experience, the power of this concept lies in its empowerment. I’ve seen clients plagued by fatigue and “brain fog” who, after years of frustration, found significant relief not through a new brain scan, but by working with a specialist to heal their gut. It reframes health from chasing symptoms to nurturing a foundational ecosystem.
This guide will demystify the gut-brain-immune axis. Whether you’re new to the idea of gut health or a professional needing the latest clinical correlations, we’ll explore the science of how these systems “talk,” why this connection is fundamental to modern medicine, and what you can do—from dietary choices to emerging therapies—to influence your own internal ecology for better health.
Background / Context: From Folklore to Frontier Science
The intuition that gut health influences overall well-being is ancient. Hippocrates is often quoted as saying, “All disease begins in the gut.” Traditional medicine systems like Ayurveda and Traditional Chinese Medicine have long emphasized digestion as the root of vitality. But for modern Western medicine, the gut was largely the domain of gastroenterologists, and its inhabitants were overlooked as mere passengers.
The turning point began with two key realizations. First, the sheer scale: the human gut microbiome contains at least as many cells as the human body itself and 150 times more genes. We are, in a genetic sense, more microbial than human. Second, the development of rapid, affordable genetic sequencing in the early 2000s allowed scientists to move from painstakingly culturing a handful of bacteria in a lab to mapping the entire complex community through its DNA—a field called metagenomics.
Landmark projects like the Human Microbiome Project (2007-2016) provided the first comprehensive reference map of microbial inhabitants in healthy individuals. This created a baseline, allowing researchers to compare and see how microbiomes deviate in disease states. Suddenly, correlations exploded: people with depression showed different microbial profiles than healthy controls; the microbiomes of children in industrialized nations, with high rates of autoimmune disease, looked radically different from those of children in traditional, agrarian societies.
The field has now moved beyond correlation to causation. Pioneering studies using germ-free mice (born and raised in sterile bubbles with no microbiome) demonstrated that transplanting gut microbes from a human with obesity, autism, or anxiety into these mice could transfer traits of the condition. This proved the microbiome wasn’t just a bystander but an active player in physiology and behavior.
The convergence of big data, advanced sequencing, and a renewed focus on holistic systems biology has propelled gut microbiome research from the fringes to the forefront of personalized medicine. For more on how data is transforming other fields, explore our analysis on global affairs and politics.
Key Concepts Defined
Navigating this field requires a clear vocabulary.
- Gut Microbiome: The entire collection of microorganisms (bacteria, archaea, fungi, viruses) and their genetic material living in your intestinal tract, primarily the large intestine or colon.
- Gut-Brain-Immune Axis: The bidirectional communication network linking the central nervous system (brain and spinal cord), the enteric nervous system (“the second brain” in the gut), the gut microbiome, and the immune system. They communicate via neural, hormonal (endocrine), and immunological pathways.
- Dysbiosis: An imbalance or maladaptation in the composition and function of the gut microbiome, often characterized by a loss of beneficial microbes, an overgrowth of potentially harmful ones, and/or a loss of overall microbial diversity. It’s considered a root driver of many chronic inflammatory conditions.
- Probiotics: Live microorganisms that, when administered in adequate amounts, confer a health benefit on the host. Found in supplements and fermented foods like yogurt, kefir, and sauerkraut.
- Prebiotics: Specific types of dietary fiber (e.g., inulin, fructooligosaccharides) that are non-digestible by humans but serve as food for beneficial gut bacteria, stimulating their growth and activity. Found in foods like garlic, onions, leeks, asparagus, and bananas.
- Postbiotics: The bioactive compounds produced by probiotic bacteria during fermentation. These include short-chain fatty acids (like butyrate), enzymes, peptides, and cell wall fragments. They are the actual mediators of many health benefits, even if the probiotic bacteria themselves are no longer alive.
- Leaky Gut (Increased Intestinal Permeability): A condition where the tight junctions between the cells lining the intestinal wall become loose, allowing undigested food particles, bacteria, and toxins to “leak” into the bloodstream, triggering systemic inflammation and immune responses.
- Short-Chain Fatty Acids (SCFAs): Powerful anti-inflammatory compounds (primarily acetate, propionate, and butyrate) produced when gut bacteria ferment dietary fiber. Butyrate, in particular, is the primary fuel source for colon cells and has profound effects on immune regulation and brain health.
- Vagus Nerve: The longest cranial nerve in the body, forming a primary physical communication highway between the gut and the brain. Gut microbes can stimulate the vagus nerve, sending signals that directly affect mood, stress response, and inflammation.
How It Works: A Step-by-Step Breakdown of the Conversation
The gut, brain, and immune system don’t just coexist; they are in constant, sophisticated dialogue. Here’s how the conversation flows.
Step 1: The Gut Microbiome Produces Signaling Molecules
As your gut bacteria break down the food you eat (especially fiber), they generate a vast array of metabolic byproducts:
- Neuroactive Compounds: Bacteria produce significant amounts of neurotransmitters like serotonin (approx. 90% of the body’s total), gamma-aminobutyric acid (GABA) (a calming neurotransmitter), and dopamine precursors.
- Short-Chain Fatty Acids (SCFAs): Butyrate, propionate, and acetate cross the intestinal lining into the bloodstream. Butyrate strengthens the gut barrier and has direct anti-inflammatory effects on immune cells throughout the body.
- Immune Modulators: Microbial components and metabolites constantly “train” and calibrate the immune system, teaching it to distinguish between friend and foe.
Step 2: Communication Via Multiple Pathways
These microbial products initiate communication through several parallel channels:
- The Neural Pathway (Vagus Nerve): Microbial signals activate nerve endings in the gut wall, which send electrical impulses up the vagus nerve to the brainstem, influencing areas that control mood (like the limbic system), stress (the HPA axis), and appetite.
- The Endocrine Pathway (Hormonal): Gut cells and microbes produce hormones and peptide signals (like peptide YY, GLP-1) that enter circulation and affect brain function and metabolism. The gut is the body’s largest endocrine organ.
- The Immune Pathway: Gut immune cells (like dendritic cells) sample microbial products and travel to other body sites, including the brain, to regulate systemic inflammation. Chronic dysbiosis can lead to a persistent state of low-grade inflammation, a key contributor to depression and neurodegenerative diseases.
- The Circulatory/Metabolic Pathway: Microbial metabolites like SCFAs and trimethylamine N-oxide (TMAO) enter the bloodstream directly, affecting the health of blood vessels, liver function, and other organs.
Step 3: The Brain and Immune System Respond
These signals elicit powerful responses:
- Brain: Altered neurotransmitter levels can directly affect mood, cognition, and stress resilience. Inflammation from the gut can compromise the blood-brain barrier and activate the brain’s immune cells (microglia), linked to brain fog, fatigue, and depression.
- Immune System: A healthy, diverse microbiome promotes the development of regulatory T-cells (Tregs), which dampen excessive immune responses and prevent autoimmunity. Dysbiosis can skew the immune system toward a pro-inflammatory state dominated by T-helper 17 (Th17) cells, implicated in autoimmune attacks.
Step 4: The Feedback Loop Completes the Circuit
The brain isn’t passive. Stress, anxiety, and sleep deprivation (brain states) alter gut motility, secretion, and permeability. This changes the gut environment, favoring some microbes over others, which in turn changes the signals sent back to the brain. It’s a continuous, dynamic loop.
Key Takeaway: Your gut bacteria act as a chemical factory. The compounds they produce travel via nerves, blood, and immune cells to instruct your brain and immune system. Your mental state and immune health then feed back to shape your gut microbiome, creating a foundational loop that determines your overall health trajectory.
Why It’s Important: The Paradigm Shift in Treating Chronic Disease
Understanding the gut-brain-immune axis is not an academic exercise; it’s transforming clinical practice and self-care for millions.
1. A Unified Theory for the Chronic Disease Epidemic
Rates of depression, anxiety, autoimmune disorders, obesity, and Alzheimer’s have soared in industrialized societies. The gut microbiome offers a compelling unifying hypothesis: the modern “Western” lifestyle—defined by diets low in fiber and high in processed foods, overuse of antibiotics, chronic stress, and reduced environmental microbial exposure—has profoundly damaged our collective microbiome, driving systemic inflammation and dysregulated immunity. Healing the gut becomes a central strategy for addressing these disparate conditions at their root.
2. Moving Beyond Symptom Suppression
Traditional medicine often excels at acute care but struggles with chronic, complex conditions, frequently resorting to lifelong medications that manage symptoms (like antidepressants or immunosuppressants) but don’t address underlying causes. The gut-axis model provides new therapeutic targets: instead of just boosting serotonin in the brain with an SSRI, we can also ask why gut serotonin production might be impaired and work to restore a microbiome that produces it optimally.
3. The Rise of Truly Personalized Medicine
Because each person’s microbiome is as unique as a fingerprint, it paves the way for ultra-personalized interventions. Microbiome testing (though still evolving) can provide insights into an individual’s microbial deficits and imbalances. This can guide targeted probiotic/prebiotic recommendations, dietary plans, and even predict responses to certain drugs, including cancer immunotherapies like checkpoint inhibitors.
4. Empowering Patient Agency
This science empowers individuals. While genetics are largely fixed, the microbiome is malleable. Daily choices about diet, sleep, stress management, and exposure to nature are direct levers everyone can pull to cultivate a healthier internal ecosystem. It shifts the narrative from passive patient to active ecosystem manager.
What I’ve found is that when patients grasp this concept, it often provides a profound “aha” moment. It connects dots between their digestive issues, low mood, and constant colds in a way that isolated diagnoses never did. This understanding fosters motivation for lifestyle change that feels meaningful and systemic.
Sustainability in the Future: Cultivating a Lifelong Internal Ecosystem
A sustainable approach to health through the gut microbiome focuses on long-term nourishment and balance, not quick fixes.
Dietary Sustainability: Fiber as Fundamental
The most powerful tool for shaping the microbiome is long-term, diverse dietary fiber intake. Fiber is the primary food (prebiotic) for beneficial bacteria that produce anti-inflammatory SCFAs. A sustainable strategy isn’t about a 30-day “gut cleanse,” but about integrating a wide variety of colorful vegetables, legumes, whole grains, nuts, and seeds into a lifelong eating pattern. Diversity of plants equals diversity of microbes.
Antibiotic Stewardship
Antibiotics are lifesaving but non-discriminatory—they wipe out beneficial bacteria along with pathogens. Their overuse (in medicine and agriculture) is a major driver of dysbiosis. A sustainable future involves using antibiotics only when absolutely necessary and actively supporting microbiome recovery afterward with evidence-based probiotics and prebiotics.
Environmental and Interpersonal Microbial Exchange
The “hygiene hypothesis” suggests that reduced exposure to diverse environmental microbes in childhood is linked to higher allergy and autoimmune rates. Sustainability means encouraging safe exposure to nature (gardening, pets, outdoor play) and recognizing that social interactions and living environments facilitate a healthy exchange of microbes that strengthen our collective resilience.
Commercial and Ethical Sustainability
The probiotic and microbiome testing market is booming, but it’s also rife with hype and unsubstantiated claims. Sustainable development requires:
- Rigorous Regulation: Ensuring probiotic supplements contain what they claim, at doses proven effective for specific conditions.
- Responsible Science Communication: Moving beyond sensational headlines (“A single bacterium causes depression!”) to communicate the complex, ecosystem-based reality.
- Data Privacy: As microbiome data is deeply personal genetic information, robust ethical frameworks must govern its testing, storage, and use.
Common Misconceptions
Let’s clear up the most frequent misunderstandings.
Misconception 1: “All probiotics are the same, and yogurt is enough.”
- Reality: Probiotics are highly strain-specific. Lactobacillus rhamnosus GG may help with traveler’s diarrhea, while Bifidobacterium longum 1714 may reduce stress. Most commercial yogurts contain strains for fermentation, not necessarily clinically studied probiotic strains at therapeutic doses. You must match the strain to the desired outcome.
Misconception 2: “A ‘leaky gut’ is a made-up condition.”
- Reality: Increased intestinal permeability is a real, measurable phenomenon in peer-reviewed research. It’s observed in conditions like Crohn’s disease, celiac disease, and severe burn trauma. The controversy lies in its proposed role in all chronic disease, which is still being researched. It is likely a key piece, but not the sole cause, for many.
Misconception 3: “You should sterilize your environment to be healthy.”
- Reality: While preventing pathogenic infection is crucial, an obsessive, sterile home environment deprives your microbiome of the diverse “training partners” it needs to develop a robust, balanced immune system. We need exposure to “old friends”—harmless microbes from people, pets, and soil.
Misconception 4: “Poop transplants are a miracle cure for everything.”
- Reality: Fecal Microbiota Transplantation (FMT) is highly effective and FDA-approved for recurrent C. difficile infection. Its use for other conditions (like obesity, autism, IBD) is experimental and carries risks, including the potential transfer of unknown pathogens or undesirable traits. It is not a DIY procedure.
Misconception 5: “More microbiome diversity is always better.”
- Reality: While generally true, it’s an oversimplification. The function of the microbiome (what it produces) is often more important than a simple species count. Someone may have lower diversity but a highly functional, stable community. The goal is a resilient, functional ecosystem, not just a long species list.
Recent Developments (2024-2025)
The field is advancing at a breathtaking pace. Here are key recent trends:
- Microbiome-Directed Therapeutics: Moving beyond generic probiotics, companies are developing consortia of specific, rationally selected bacterial strains designed to perform missing metabolic functions. For example, Seres Therapeutics’ SER-109 (a purified bacterial spore product) recently gained FDA approval for preventing recurrent C. diff, marking a landmark for microbiome drugs.
- The “Viral” Side: The Virome and Phage Therapy: Attention is expanding beyond bacteria to the gut virome—the community of viruses, particularly bacteriophages that infect bacteria. Phage therapy is being explored to precisely target pathogenic bacteria without harming beneficial ones, offering a potential alternative to broad-spectrum antibiotics.
- AI-Powered Microbiome Analysis: Machine learning algorithms are now being used to analyze complex microbiome data, identifying specific microbial signatures that predict disease risk, drug response (especially in oncology), and optimal dietary interventions for individuals.
- The Gut-Brain Connection in Neurodegeneration: A landmark study in Nature in late 2024 provided stronger evidence that specific gut microbes in older adults are associated with the accumulation of amyloid proteins, a hallmark of Alzheimer’s disease, potentially via increased production of pro-inflammatory metabolites.
- Personalized Nutrition 2.0: Companies like ZOE and others are offering large-scale personalized nutrition programs that combine continuous glucose monitoring with microbiome and blood fat testing, providing real-time, individualized feedback on how a person’s unique gut microbes respond to specific foods.
Success Stories and Real-Life Examples

Case Study 1: Inflammatory Bowel Disease (IBD)
- Challenge: A patient with ulcerative colitis struggles with frequent flares despite standard immunosuppressant medications, experiencing abdominal pain, fatigue, and depression.
- Solution: As part of a comprehensive care plan at a specialized clinic, the patient undergoes microbiome analysis, revealing very low levels of SCFA-producing bacteria and high levels of a pro-inflammatory strain. The treatment plan includes a targeted prebiotic fiber protocol (like resistant starch) to feed beneficial bacteria, a specific probiotic strain (E. coli Nissle 1917) with evidence in maintaining remission, and dietary counseling to reduce inflammatory triggers.
- Outcome: Over 6-9 months, the patient experiences longer remission periods, reduced systemic inflammation markers, and an improved sense of well-being and energy, illustrating the gut-brain-immune connection directly.
Case Study 2: Major Depressive Disorder
- Challenge: A patient with treatment-resistant depression finds minimal benefit from multiple antidepressants (SSRIs/SNRIs).
- Solution: The psychiatrist, aware of the gut-brain research, collaborates with a registered dietitian. They implement a “psychobiotic” dietary intervention focused on increasing prebiotic fiber and fermented foods, while also adding a probiotic supplement containing clinically studied strains like Lactobacillus helveticus and Bifidobacterium longum. They also address sleep and stress, which impact gut permeability.
- Outcome: While not a standalone cure, the patient reports a noticeable improvement in mood stability, reduced anxiety, and better sleep after 12 weeks. This allows the antidepressant medication to work more effectively, showcasing an adjunctive, systems-based approach.
Case Study 3: Cancer Immunotherapy Response
- Challenge: Two similar patients with metastatic melanoma receive the same immunotherapy drug (a PD-1 inhibitor). One responds spectacularly, the other does not.
- Solution: Research led by the MD Anderson Cancer Center and others has identified that responders have a significantly different and more diverse gut microbiome, enriched with bacteria like Faecalibacterium and Bifidobacterium. These bacteria are thought to prime the immune system for a more effective anti-tumor response.
- Outcome: This has led to active clinical trials where non-responders receive FMT from responders alongside immunotherapy, with some early studies showing remarkable success in converting non-responders. This is a powerful example of the microbiome dictating treatment efficacy.
Conclusion and Key Takeaways
The discovery of the gut-brain-immune axis represents one of the most significant medical revelations of the 21st century. It dismantles the outdated view of body systems operating in isolation and replaces it with a dynamic, interconnected model of health centered on our internal microbial ecosystem.
To integrate this knowledge into your life and practice, remember:
- You Are an Ecosystem: Health is the product of a symbiotic relationship between your human cells and trillions of microbial partners. Nurturing this partnership is foundational.
- Fiber is Fertilizer: Diverse dietary fiber is the most powerful, evidence-based tool you have to cultivate a beneficial microbiome. Aim for 30+ different plant foods weekly.
- Think in Systems, Not Symptoms: Unexplained fatigue, low mood, and recurrent inflammation may share a common root in gut dysbiosis. Addressing gut health can have cascading positive effects.
- The Future is Personalized and Microbial: Medicine is moving towards interventions—from probiotics to FMT—precisely tailored to an individual’s microbial makeup to treat and prevent disease.
- Lifestyle is Primary Care: Daily decisions about food, sleep, stress, and environment are direct levers for shaping your microbiome and, by extension, your long-term brain and immune health.
This is not a passing fad but a permanent expansion of our understanding of human biology. By embracing our role as stewards of this inner world, we open new, empowering pathways to resilience and vitality.
Frequently Asked Questions (FAQs)
1. What’s the single best thing I can do for my gut health?
Increase the diversity and amount of plant-based fibers in your diet. Different bacteria thrive on different fibers, so eating a wide variety of vegetables, fruits, legumes, nuts, seeds, and whole grains is the most effective strategy.
2. Are expensive microbiome tests worth it?
For the average healthy person, they are often more interesting than clinically actionable. Their interpretation is complex, and clear “ideal” benchmarks are still being defined. They can be more useful for individuals with specific, stubborn health issues under the guidance of a knowledgeable healthcare practitioner who can translate the results into a targeted plan.
3. Can improving my gut health help with weight loss?
It can be a significant factor. A dysbiotic microbiome may be more efficient at extracting calories from food, promote low-grade inflammation that drives insulin resistance, and affect hormones that regulate hunger and satiety (like GLP-1). Fixing the microbiome supports, but does not replace, other healthy weight management strategies.
4. How long does it take to change your microbiome?
Dietary changes can induce shifts in microbial populations within 24-48 hours, but these are often transient. To create lasting, stable changes in your core microbiome, consistent dietary and lifestyle habits over several months to a year are typically needed.
5. Is kombucha really that good for you?
Kombucha is a fermented tea that can contain live probiotics, organic acids, and antioxidants. It may contribute to microbial diversity for some people. However, it’s not a magic elixir, and many commercial varieties are high in added sugar. It’s best viewed as one optional component of a gut-healthy diet, not a cornerstone.
6. What’s the difference between a probiotic and a postbiotic?
A probiotic is the live microorganism itself (the “worker bee”). A postbiotic is the beneficial compound the microbe produces (the “honey”). Postbiotic supplements contain these compounds directly and are stable, don’t require refrigeration, and may be effective even in people whose gut conditions might kill live probiotics.
7. Can stress really damage my gut?
Absolutely. Chronic stress alters gut motility, increases permeability (“leaky gut”), and changes the gut environment in ways that can favor less beneficial microbes. This is a prime example of the brain-to-gut side of the axis.
8. Should I take a probiotic during/after antibiotics?
Yes, but timing and strain matter. Take the probiotic 2-3 hours apart from the antibiotic dose. Choose a well-researched, broad-spectrum probiotic with strains like Lactobacillus rhamnosus GG and Saccharomyces boulardii (a beneficial yeast), which have good evidence for preventing antibiotic-associated diarrhea. Continue for at least a few weeks after the antibiotic course.
9. Are there any risks to taking probiotics?
For most healthy people, they are very safe. However, in critically ill individuals, those with severe immune compromise, or those with central venous catheters, there is a rare risk of systemic infection from the probiotic. Always consult your doctor if you have serious underlying health conditions.
10. What is the “psychobiotic” diet?
While not a rigid diet, it’s an eating pattern emphasizing foods that support a mood-boosting microbiome: ample prebiotic fibers, polyphenol-rich foods (berries, dark chocolate, green tea), fermented foods, and healthy fats (like omega-3s from fish), while minimizing ultra-processed foods, artificial sweeteners, and excessive saturated fat.
11. Does the microbiome change with age?
Yes. Older adults often show a decrease in microbial diversity and a shift in populations, which may contribute to age-related inflammation (“inflammaging”) and frailty. This makes lifelong dietary fiber intake even more critical for healthy aging.
12. Can children’s gut health affect their development?
Critically so. Early-life microbiome establishment is linked to immune system development, risk for allergies and asthma, and even neurodevelopmental outcomes. Mode of delivery (vaginal vs. C-section), breastfeeding, and early diet play monumental roles in shaping this initial microbial community.
13. What’s the connection with autoimmune diseases?
Dysbiosis can break immune tolerance—the immune system’s ability to ignore our own tissues. A “leaky gut” may allow bacterial fragments into the bloodstream that molecularly mimic human tissues (a theory called molecular mimicry), tricking the immune system into attacking the body. This is a leading hypothesis for diseases like rheumatoid arthritis and multiple sclerosis.
14. Are all fermented foods probiotic?
No. For a fermented food to be considered probiotic, it must contain a defined, live microorganism in an adequate dose that survives digestion and confers a proven health benefit. Many traditionally fermented foods (like sauerkraut, kimchi, and some yogurts) contain live cultures but may not have been studied to meet this strict definition. They are still excellent for gut health.
15. How does exercise affect the gut microbiome?
Regular, moderate exercise is associated with increased microbial diversity and higher levels of beneficial SCFA-producing bacteria. It may also improve gut barrier function. However, extreme, prolonged endurance exercise can temporarily increase gut permeability and cause distress.
16. Is there a link to skin conditions like acne or eczema?
Yes, via the gut-skin axis. Systemic inflammation and immune dysregulation stemming from the gut can manifest in the skin. Many patients with acne or eczema see improvements when they address gut health through diet and probiotics.
17. What are “microbial metabolites” and why are they important?
They are the small molecules produced by gut bacteria as they metabolize food. They include the SCFAs, neurotransmitters, vitamins (B and K), and various other compounds. These metabolites are the primary mechanism by which the microbiome exerts its vast influence on the host—they are the language of the axis.
18. Can I damage my microbiome forever?
The microbiome is generally resilient but can be permanently altered by major early-life events (like C-section birth without microbial seeding, or lack of breastfeeding) or repeated, severe insults (like multiple broad-spectrum antibiotic courses). However, even in these cases, strategic, long-term lifestyle interventions can often push it toward a healthier, functional state.
19. What role do polyphenols play?
Polyphenols (in colorful berries, red wine, tea, dark chocolate) are plant compounds that are poorly digested by us but are excellent prebiotics for beneficial bacteria. They also have direct anti-inflammatory effects. Think of them as high-quality “super-fuel” for your good gut bugs.
20. Where can I find a doctor who understands this?
Look for functional medicine doctors, integrative gastroenterologists, or naturopathic physicians who emphasize root-cause medicine. Some forward-thinking psychiatrists (sometimes called “nutritional psychiatrists”) and dietitians specializing in gut health are also excellent resources. Always verify credentials.
About the Author
This guide was written by a science communicator and health educator with a background in molecular biology and a passion for translating complex medical research into actionable insights. Having worked in clinical research coordination and patient advocacy, the author has witnessed firsthand the frustration of chronic illness and the transformative potential of systems-based approaches like microbiome medicine. They are dedicated to providing evidence-based, nuanced information that empowers individuals to partner more effectively with their healthcare providers. For more explanations of complex topics, visit our Explained section.
Free Resources to Continue Your Learning
- The American Gastroenterological Association (AGA) Gut Microbiome Center: Provides patient-friendly summaries of the latest research.
- The International Scientific Association for Probiotics and Prebiotics (ISAPP): A non-profit that publishes reliable, scientist-vetted infographics and fact sheets on probiotics, prebiotics, and postbiotics.
- PubMed Central (PMC): A free archive of biomedical literature. Search for terms like “gut-brain axis review” or “microbiome and [your condition of interest]” to access original research papers.
- The ZOE Science & Nutrition Podcast: Features leading microbiome researchers and breaks down new studies in an accessible way.
Discussion
The gut microbiome revolution raises as many questions as it answers. We invite you to join the conversation.
- Has learning about the gut-brain-immune axis changed how you view your own health symptoms?
- What dietary or lifestyle change have you found most impactful for your digestion and overall well-being?
- What concerns or hopes do you have about the future of microbiome-based medicine?
Share your thoughts and questions. For insights on other global challenges and systemic thinking, you might find our coverage on global supply chain management offers a fascinating parallel in understanding complex, interconnected systems.



